Cavitation Bubbles: How “Bursting” Micro-explosions Can Create Better Chitosan

Pixabay
Date of publication:
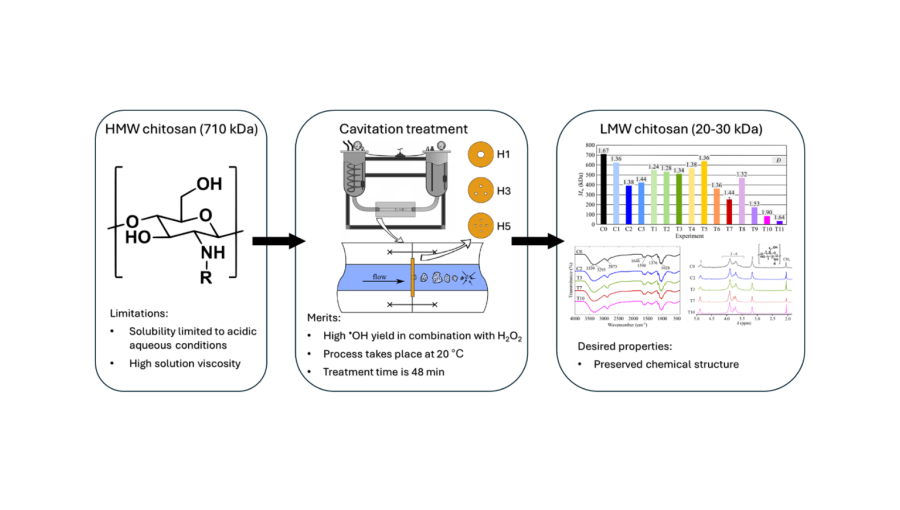
Researchers from the Faculty of Mechanical Engineering UL and the Institute of Chemistry have developed a new, faster, and more environmentally friendly method for preparing low molecular weight chitosan. In their work, they utilized cavitation—a clean, fast, and efficient technology that could replace some less sustainable existing methods. This breakthrough opens the way for the development of a new generation of materials that could be used in medicine, pharmacy, food applications, industry, and environmental solutions.
In this study, the researchers aimed to use cavitation and the addition of an oxidant—hydrogen peroxide—to shorten long chitosan chains without damaging its backbone structure. They investigated several parameters that influence the degradation process, including cavitation intensity, hydrogen peroxide concentration, and sample preparation. The researchers successfully reduced the weight-average molecular weight of chitosan by as much as 97.2%. Importantly, its backbone structure remained unchanged, meaning that the material retains all its key functional properties.
These findings are important because conventional methods for chitosan degradation are often slow, require high temperatures, or involve aggressive chemicals that can damage the material’s structure. The cavitation-based method, however, is faster, more energy-efficient, and significantly more environmentally friendly. It is also suitable for industrial application, meaning it could be used in the future for scalable and sustainable production of bioactive materials.
What is chitosan?
Chitosan is a polysaccharide—a complex carbohydrate composed of multiple monosaccharide units such as glucose. It is derived from chitin, which is the main component of the exoskeletons of crustaceans and shellfish. When these long chains are shortened, low molecular weight chitosan is obtained, which is more soluble, biologically active, and more suitable for use in medical materials. Due to its properties, it is particularly interesting for drug production, the preparation of bioactive surfaces, the development of biocompatible materials, and various environmental applications, including water purification.
What is cavitation?
Cavitation occurs due to a sudden local drop in pressure, leading to the formation of small vapor bubbles in a liquid. When these cavitation bubbles collapse, the released energy is concentrated in an extremely confined area and within a very short time, which can be compared to micro-explosions. This results in intense pressure fluctuations, the formation of microjets, and locally extreme temperatures, which can cleave molecular bonds or trigger various chemical reactions.
The study thus confirms that cavitation bubbles, if properly controlled, are an extremely promising tool for the degradation of polysaccharides such as chitosan. Compared to chemical and enzymatic methods, they represent a clean, fast, and efficient technology. Moreover, they open the way for the development of a new generation of materials that could be used in medicine, pharmacy, food applications, industry, and environmental solutions.
The research was carried out by an interdisciplinary team of experts from the Faculty of Mechanical Engineering, the Institute of Chemistry, and the Faculty of Pharmacy, including Andraž Zupanc, Jernej Ortar, David Pahovnik, Nataša Zabukovec Logar, Blaž Stres, Alenka Šmid, Mojca Zupanc, Martin Petkovšek, and Ema Žagar. The results were published in the scientific journal Ultrasonics Sonochemistry.